
News
From Front-runner to Comprehensive Layout! Sino Biopharm Accelerates Development of TQC3721 Dry Powder Inhaler and Combination Formulations, Solidifying Its Therapeutic Position in the Respiratory Field
Release Date: 2026-03-19
Recently, the results of the Phase II clinical trial (PACER-II) of TQC3721 suspension, a PDE3/4 inhibitor independently developed by CTTQ Pharma, a core member company of Sino Biopharm (1177.HK), for Chronic Obstructive Pulmonary Disease (COPD), were officially published in the renowned international respiratory journal CHEST. PACER means Front-runner in a marathon. The development progress of TQC3721 suspension is not only the fastest among domestic products, but the topline data from its first Phase III clinical trial for the maintenance treatment of COPD is also expected to be read out by the end of 2026. The combination formulation of TQC3721 inhalation powder is also being developed concurrently, which will further solidify the Group's important therapeutic position in the respiratory field.
.jpg/3_19(1)__600x287.jpg)
Study Background
COPD is a lung disorder characterized by chronic respiratory symptoms (dyspnoea, cough, sputum production), caused by chronic airway inflammation and persistent airflow obstruction. COPD is one of the top three causes of death worldwide, affecting over 391 million people. Despite the availability of various therapeutic drugs, COPD patients still face a shortage of medications when treatment escalation is needed.
Current drug therapies for COPD, such as long-acting muscarinic antagonists (LAMAs) and long-acting β2-agonists (LABAs), aim to improve lung function but have limited efficacy in controlling airway inflammation. Inhaled corticosteroids (ICS) are the primary treatment for chronic inflammatory airway diseases, and their combination with LABA or LAMA has been shown to reduce COPD exacerbations. However, ICS has limited benefits for COPD patients with low blood eosinophil counts and increases the risk of pneumonia. Therefore, it is necessary to develop drugs that can simultaneously alleviate airway inflammation and airflow obstruction.
Phosphodiesterases (PDEs) are an enzyme superfamily responsible for regulating cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) by breaking them down into inactive forms. PDE3 is highly expressed in airway smooth muscle, while PDE4 is expressed in almost all inflammatory cells. As a dual-target PDE3/4 inhibitor, TQC3721 suspension controls disease progression through a dual mechanism of improving ventilation by relaxing airway smooth muscle and inhibiting inflammation.
Study Design
PACER-II was a Phase II, multicenter, randomized, double-blind, placebo-controlled trial led by Professor Weimin Li of West China Hospital, Sichuan University, and jointly completed by 27 research centers in China. It aimed to evaluate the preliminary efficacy and safety of TQC3721 suspension in COPD patients receiving background therapy with a single or dual bronchodilator. A total of 240 patients with moderate-to-severe COPD were enrolled, and all subjects were receiving maintenance therapy with LAMA, LABA/LAMA, or triple therapy.
204 eligible subjects were randomly assigned in a 1:1:1 ratio to receive treatment with 3 mg TQC3721, 6 mg TQC3721, or placebo, twice daily for a 4-week treatment period. At baseline, 55.4% and 44.6% of patients had lung function severity corresponding to GOLD stage 2 and 3, respectively. Based on different background therapies, 28.8% and 71.2% of patients were using LAMA and LABA/LAMA as background therapy, respectively. The primary endpoint was the change from baseline in peak forced expiratory volume in 1 second (FEV1) during the 4-week treatment period.
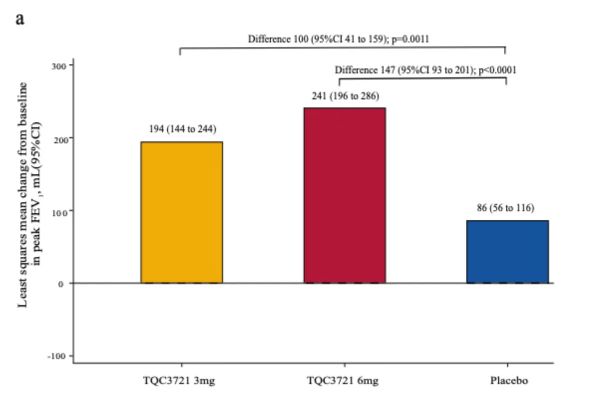
Study Results
At week 4 of TQC3721 treatment, the change from baseline in peak FEV1 showed significant improvement (Figure a). The mean increase in peak FEV1 was 194 mL (95% CI: 144-244, P < 0.0001) in the 3 mg dose group and 241 mL (95% CI: 196-286, P < 0.0001) in the 6 mg dose group. Compared to placebo, both the 3 mg and 6 mg TQC3721 groups were significantly superior, with differences of 100 mL (95% CI: 41-159, P = 0.0011) and 147 mL (95% CI: 93-201, P < 0.0001), respectively.
The 12-hour post-dose FEV1 curve at week 4 showed that the 6 mg dose of TQC3721 had a better bronchodilator effect than the 3 mg dose, suggesting a significant dose-response relationship for the treatment effect (Figure b). In addition, the change from baseline in trough FEV1 also showed a clear dose-response relationship (Figure c).
(7).png/WPS%E5%9B%BE%E7%89%87(1)(7)__586x385.png)
.png/c(1)__600x368.png)
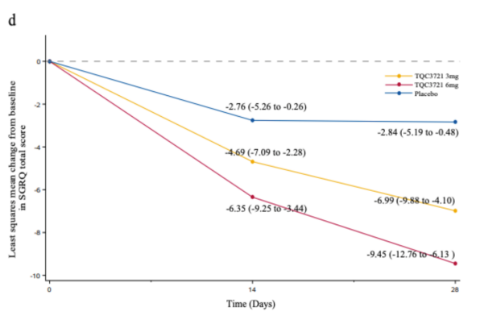
The St. George's Respiratory Questionnaire (SGRQ) scores further demonstrated a significant improvement in patients' symptoms and quality of life at week 4 of TQC3721 treatment. Compared to the placebo group, the mean score reduction was 3.56 (95% CI: -6.52 to -0.61, P = 0.0185) in the 3 mg dose group and 5.09 (95% CI: -8.44 to -1.74, P = 0.0031) in the 6 mg dose group (Figure d). The improvement from baseline in mean SGRQ scores for both the 3 mg dose group (-6.99 [-9.88, -4.10], P < 0.0001) and the 6 mg dose group (-9.45 [-12.76, -6.13], P < 0.0001) exceeded the minimal clinically important difference threshold of -4 points. Furthermore, the proportions of patients who met the definition of a treatment responder were 51.85% and 59.49% in the 3 mg and 6 mg dose groups, respectively, both higher than the 33.75% in the placebo group (P = 0.0040).
Significant clinical benefits were also observed in the subgroup analyses of LAMA and LABA/LAMA background therapies, as well as in the GOLD 2 and GOLD 3 subgroups.
Safety
In this study, inhaled TQC3721 suspension demonstrated a favorable safety profile. The overall profile and incidence of adverse events were similar between the TQC3721 groups and the placebo group.
This study was named PACER (Front-runner), symbolizing its role in leading innovative drug R&D in the field of COPD. Based on the positive progress of the Phase II study, a pivotal Phase III validation study of TQC3721 suspension for the maintenance treatment of COPD is underway (NCT07147946). In July 2025, regulatory authorities approved the breakthrough therapy application for TQC3721 suspension in the treatment of COPD, and the first Phase III trial is expected to have topline data by the end of 2026.
In addition to the inhalation suspension, the TQC3721 inhalation powder has also entered the Phase II clinical trial stage, with topline data expected in mid-2026. At that time, communication with regulatory authorities regarding the initiation of pivotal validation studies will take place. The company is also concurrently advancing the development of a combination formulation of the TQC3721 inhalation powder to further solidify the position of PDE3/4 inhibitors in the treatment of COPD.
Disclaimer:
1. This press release is intended to facilitate the communication and exchange of medical information and is for reference by healthcare professionals only. It is not for advertising purposes.
2. The company does not recommend any drugs and/or indications.
3. The information contained in this press release is for reference only and cannot replace professional medical guidance in any way, nor should it be considered as a diagnosis or treatment recommendation. If you wish to understand specific information about disease diagnosis and treatment, please follow the advice or guidance of a doctor or other healthcare professional.
Forward-looking Statements:
This press release contains certain forward-looking statements, including statements regarding the clinical development plan, expected clinical benefits and advantages, commercialization outlook, the likelihood of clinical benefit for patients, and potential commercial opportunities for [TQC3721]. Words such as "expect", "believe", "continue", "may", "estimate", "hope", "intend", "plan", "potential", "predict", "project", "should", "will", "propose", and similar expressions are intended to identify forward-looking statements, but not all forward-looking statements contain these identifying words. These forward-looking statements are predictions or expectations made by the company based on currently available data and information, and actual results may differ materially from these forward-looking statements due to uncertainties or risks such as policy, R&D, market, and regulatory factors. Current or potential investors are advised to carefully consider the potential risks and should not place undue reliance on the forward-looking statements in this press release, which contain information only as of the date of this press release. Unless required by law, the company undertakes no obligation to update or revise any forward-looking statements in this press release as a result of new information, future events, or other circumstances.
- Previous:Breaking Medication Restrictions! Sino Biopharm's CCR8+PD-1 Combination Formulation TQB6457 Receives First Clinical Trial Approval
- Next:Eric Tse Attends Yabuli Entrepreneurs Forum: Breaking the Boundaries of Perception, Technology, and Cooperation to Explore the Path of Innovation and Establishment for Chinese Pharmaceutical Companies




