
News
Authoritative Citeline Report: Sino Biopharmaceutical Ranks 14th Globally in Pipeline Scale, Demonstrating "Impressive Achievement"
Release Date: 2026-04-03
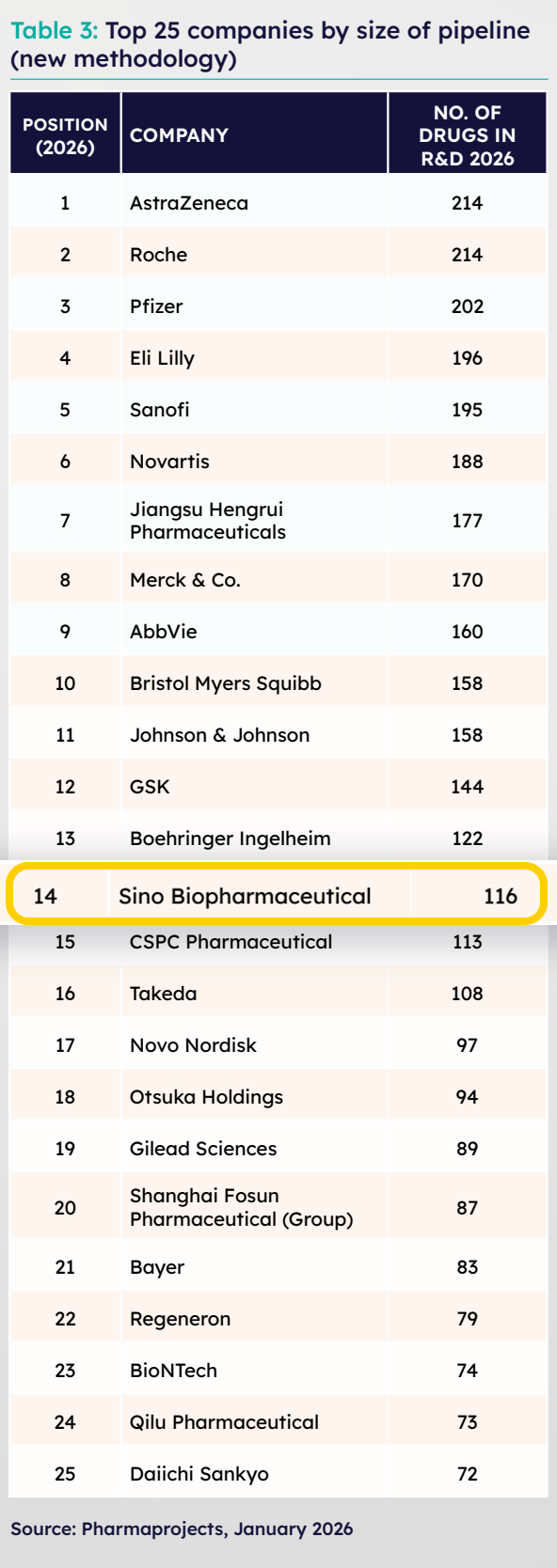
Recently, the internationally renowned consulting firm Citeline released the "Pharma R&D Annual Review 2026" and introduced a new, more precise analytical method to select the "Top 25 Global Pharmaceutical Companies by Pipeline Scale". The report shows that Sino Biopharmaceutical (1177.HK) ranks 14th globally in R&D pipeline scale, an improvement of one position compared to traditional statistical methods. The number of active drugs in development has reached 116, further highlighting that the "Impressive Achievement" of its pipeline is at the forefront among top global pharmaceutical companies.
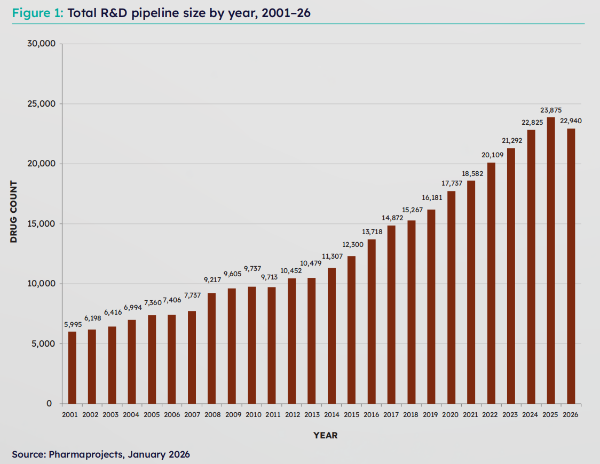
The Citeline "Pharma R&D Annual Review" series has been published continuously since 1993 and has become an authoritative report in the global pharmaceutical R&D field. Data from 2026 shows that the total number of drugs in development worldwide (including preclinical to marketed stages) was 22,940, a year-on-year decrease of 3.92%. This marks the first decline in over 20 years, and this pullback reflects a shift in the industry from "quantitative expansion" to "pursuing high quality and core competitiveness". With 1,373 new drug R&D initiations, China has become the world's second-largest contributor to R&D, second only to the United States.
Notably, Citeline adopted a new analytical method this year to more accurately reflects a company's pipeline activity, avoiding errors caused by past collaboration records or inactive associations. Based on the new statistical methodology, Sino Biopharmaceutical's R&D pipeline scale ranks 14th globally, with 116 active drugs in development. After removing data from "inactive associated pipelines," the statistical values for most cases showed a significant decline. However, Sino Biopharmaceutical's global ranking rose, directly reflecting the extremely high "Impressive Achievement" of its pipeline and indirectly demonstrating the company's robustness in actual R&D execution.
After the adjustment, more Chinese pharmaceutical companies appeared on the list of the top 25 global pipelines, further proving that the R&D pipelines of Chinese pharmaceutical companies are not only large in scale but also demonstrate a stronger "hands-on" attribute in the global R&D race.
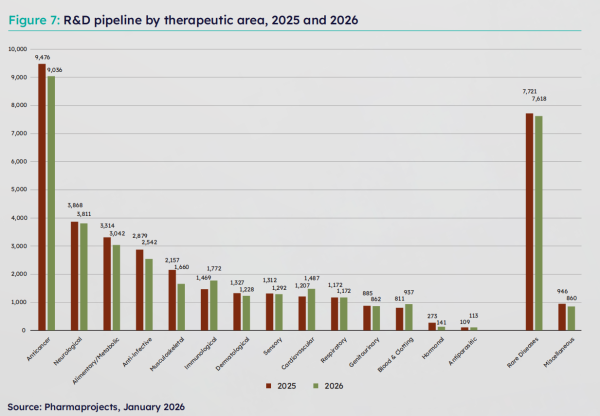
In terms of therapeutic areas, the oncology field remains the core focus of the industry. The report shows that in 2025, 4,488 new drug candidates were added globally, with the oncology field contributing 38.6%, once again becoming the most intensive area for R&D. In terms of pipeline scale, although there was an overall contraction, areas such as immunotherapy, cardiovascular disorders, and blood/coagulation systems grew against the trend. Among them, the number of candidate drugs in the immunotherapy field surged by 20.6%, becoming the standout performer.
As one of Sino Biopharmaceutical's four core therapeutic areas, the Group has a total of 39 Class 1 innovative drugs in the oncology field at the clinical development stage or beyond (as of the 2025 performance reporting period). In terms of independent R&D, the indications for the core product Anlotinib Hydrochloride (Fukewei®) in combination with Benmelstobart Antibody (Andewei®) and Penpulimab (Annike®) continue to expand for first-line/later-line treatment in areas such as lung cancer, soft tissue sarcoma, renal cell carcinoma, endometrial cancer, hepatocellular carcinoma, and thyroid cancer. In 2025, the Group further strengthened its R&D capabilities in cutting-edge molecular types and immuno-oncology through the acquisition of LaNova Medicines, and gained several "first-in-class (FIC)" candidate drugs. Among them, LM-108 (a CCR8 monoclonal antibody) is expected to become the "Anlotinib" of the digestive tract cancer field, and the data for LM-302 (a CLDN18.2 ADC) fully support its potential to be "best-in-class (BIC)". In early 2026, the acquisition of Hygieia further enhanced the Group's layout in major chronic disease areas such as cardiovascular and cerebrovascular, metabolic, and neurological diseases. Kylo-11, the world's first clinically validated siRNA drug with annual dosing potential, is currently undergoing Phase II clinical trials in both China and the US and is attracting significant attention.
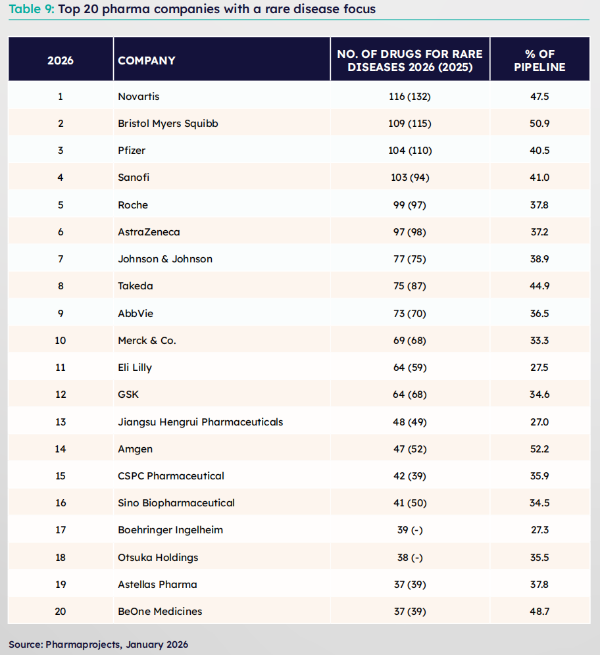
In the rare disease field, a track long dominated by multinational corporations (MNCs), Sino Biopharmaceutical ranks among the global top 20 with a pipeline scale of 41 assets in the rare disease area. In the field of blood disorders, Chia Tai Tianqing's independently developed Recombinant Human Coagulation Factor VIII for Injection (Anhengji®) and Recombinant Human Coagulation Factor VIIa N01 for Injection (Anqixin®) jointly build a solid protective system covering "routine treatment + inhibitor management" for hemophilia; after the approval and marketing of Ruxolitinib (Anxu®), a licensing collaboration worth $1.53 billion was reached with Sanofi, setting a record for the largest transaction in China's transplantation field.
Behind the impressive performance is the company's sustained high proportion of R&D investment for many consecutive years. In 2025, the Group's full-year R&D expenses reached RMB 5.87 billion, accounting for 18.4% of its revenue. Currently, Sino Biopharmaceutical has established R&D centers in Beijing, Shanghai, Guangzhou, Jiangsu, and other locations, and has successfully built ten core technology platforms covering small molecules, monoclonal antibodies, bispecific/multispecific antibodies, ADCs, siRNA, and PROTAC, efficiently advancing more than 130 clinical studies throughout the year. In the past three years (2023-2025), the Group has received cumulative approval for 16 innovative products (including 7 national Class 1 innovative drugs).
Focusing on broader clinical needs, Sino Biopharmaceutical has laid out a pipeline matrix with clear tiers and ample reserves.As multiple blockbuster products with global FIC/BIC potential successively enter the commercialization period, Sino Biopharmaceutical is set to usher in a new round of explosive growth in the next three years, fully embarking on a new era of international expansion and high-quality development.




