
News
Policy Empowers Innovation Implementation! Naldemedine Launched in the Guangdong-Hong Kong-Macao Greater Bay Area, Accelerating Benefits for Domestic OIC Patients
Release Date: 2026-04-09
Opioid-induced constipation (OIC) is one of the most common symptoms in cancer patients, affecting approximately 60%-90% of cancer patients using opioids for pain relief[1]. On April 7, Naldemedine, a treatment for OIC exclusively introduced by Chia Tai Tianqing, a core enterprise of Sino Biopharmaceutical (1177.HK), was officially approved by the Guangdong Provincial Medical Products Administration for launch in the Guangdong-Hong Kong-Macao Greater Bay Area.
In March this year, the Guangzhou Municipal Health Commission issued the Notice on Ten Measures to Promote the In-depth Implementation of the "Hong Kong and Macao Medicine and Medical Device Access" Policy, marking a new stage for the policy's transition from exploration to large-scale implementation. Naldemedine has also become one of the first innovative drugs to enter the Greater Bay Area through the "Hong Kong and Macao Medicine and Medical Device Access" since the new policy was announced.
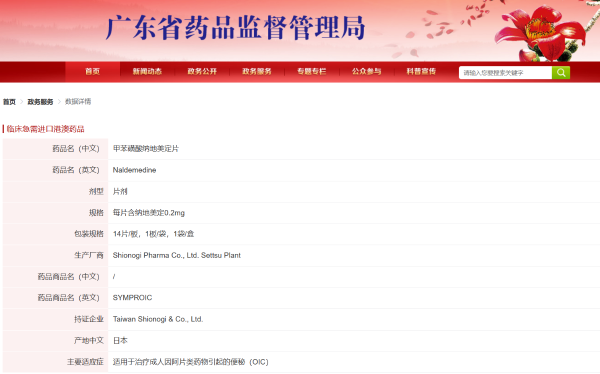
Naldemedine, developed by Shionogi & Co., Ltd., is the world's first approved oral, peripherally-acting μ-opioid receptor antagonist. It directly restores normal intestinal peristalsis and fluid secretion by blocking the binding of opiates to μ-receptors in the gut, thus reversing the pathological process of OIC at its source[2]. A 2-week, randomized, double-blind, placebo-controlled Phase III trial showed that the median time to the first spontaneous bowel movement (SBM) in OIC patients after the first dose of Naldemedine was 4.67 hours, significantly better than the placebo group (26.58 hours, P<0.0001). The SBM response rate in the Naldemedine treatment group was 71.1%, significantly higher than the placebo group (34.4%, P<0.0001), demonstrating that 0.2 mg of Naldemedine can effectively control OIC and provide sustained, stable improvement in intestinal function.
The incidence of pain in advanced cancer patients can reach 60%-80%, with about one-third of these patients experiencing moderate to severe pain. Opioids are the primary method for treating chronic pain in cancer patients, but 60%-90% of patients experience OIC symptoms, which severely impact their quality of life and treatment adherence. How to effectively treat OIC while balancing analgesia and the occurrence of adverse reactions has become one of the urgent clinical problems to be solved. Currently, laxatives commonly used in clinical practice to treat OIC may cause gastrointestinal adverse reactions such as nausea, vomiting, diarrhea, and abdominal pain, and they only treat the symptoms, not the root cause. Although traditional opioid receptor antagonists like naloxone can alleviate constipation by weakening the gastrointestinal effects of opiates, they may also reduce the analgesic effect of opiates by crossing the blood-brain barrier[3]. In contrast, Naldemedine selectively acts on peripheral μ-opioid receptors in the gut, carries no risk of electrolyte imbalance, is suitable for the long-term medication needs of OIC patients, and barely enters the central nervous system, with no observed impact on the analgesic effects of opioids.
Currently, Naldemedine has been approved for marketing in the United States, the European Union, Japan, Taiwan, China, as well as Hong Kong and Macao, China, and is recommended by the American Gastroenterological Association (AGA) guidelines and the European Society for Medical Oncology (ESMO) guidelines for the treatment of OIC[4,5]. In January 2025, Chia Tai Tianqing signed an exclusive marketing agreement with Shionogi & Co., Ltd., and was granted the exclusive right to market Naldemedine in mainland China. Previously, the product had been introduced to Hainan Province as a clinically urgently needed drug through the policy of the Boao Lecheng International Medical Tourism Pilot Zone; in May 2025, its New Drug Application in mainland China was accepted by the NMPA; this time, the approval of Naldemedine for marketing in the Greater Bay Area will significantly improve drug accessibility and address the needs of more OIC patients.
As the nationwide launch process of Naldemedine advances, Chia Tai Tianqing will leverage its commercialization capabilities to bring this globally innovative drug to more OIC patients, helping to enhance the standard of supportive care in oncology in China.
References:
[1] Porta-Sales J, Nabal-Vicuna M, Vallano A, et al.Have we improved pain control in cancer patients?A multicen-ter study of ambulatory and hospitalized cancer patients[J].J Palliat Med, 2015,18(11):923-932.
[2]John M, Edward J, et al.Peripherally Acting u-Opioid Receptor Antagonists for theTreatment of Opioid-Related Side Effects: Mechanism of Action and Clinical Implica-tions.
[3] Wang Y, Jiang K. Pathological mechanism and treatment progress of constipation induced by opioids[J]. Chinese Journal of Clinical Oncology, 2021, 48(16):852-857.
[4] Seth D.Crockett, Katarina B.Greer, Joel J.Heidelbaugh,et al.American Gastroenterological Association Institute Guideline on the Medical Management of Opioid-Induced Constipation.Gastroenterology 2019;156:218–226.
[5]N.Katakami, T.Harada2, T.Murata,et al.Diagnosis, assessment and management of constipation in advanced cancer: ESMO Clinical Practice Guidelines.Annals of Oncology 29(Supplement4):iv111-iv125, 2018.




