
News
Leading the Next Generation of ADC Innovation! LaNova Medicines Showcases Two Differentiated Innovative Products at AACR 2026
Release Date: 2026-04-22

From April 17 to 22, local time, the 2026 American Association for Cancer Research (AACR) Annual Meeting was held in San Diego. As one of the most influential academic events in the global oncology research field, LaNova Medicines, a wholly-owned subsidiary of Sino Biopharmaceutical (1177.HK), disclosed for the first time key preclinical research data for two globally competitive next-generation antibody-drug conjugates (ADCs) - LM-364TME and LM-338. These two investigational drugs focus on tumor microenvironment (TME) precision activation and a novel, highly tumor-specific target, respectively, showcasing LaNova Medicines's differentiated innovation capabilities in the next-generation ADC field.
LM-364TME: Breaking the "On-Target/Off-Tumor" Bottleneck
(8).png/WPS%E5%9B%BE%E7%89%87(1)(8)__600x191.png)
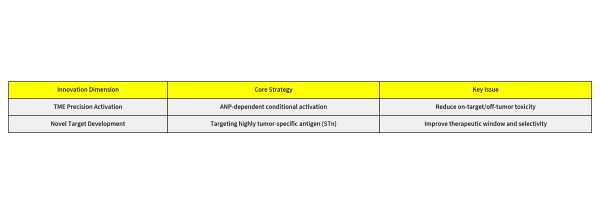
Toxicity from ADCs that do not reach the target lesion or tissue can be classified as "on-target" or "off-target" toxicity, where target-mediated toxicity occurs through the binding of the ADC to antigen proteins on healthy cells. Each component of an ADC, including the antibody, linker, and payload, can influence the degree of ADC-induced toxicity. Nectin-4 is a clinically validated ADC target that is highly expressed in various solid tumors such as urothelial carcinoma and triple-negative breast cancer. However, its low-level expression in normal tissues often leads to dose-limiting toxicities like rash and neurotoxicity, becoming a bottleneck in clinical development.
To address this key challenge, LaNova Medicines has successfully developed a next-generation Nectin-4 ADC, LM-364TME, based on its proprietary tumor microenvironment-responsive ADC platform. This molecule employs an adenine nucleotide (ANP)-dependent binding mechanism: since the ANP concentration in the tumor microenvironment (micromolar range) is significantly higher than in normal tissues (nanomolar range), LM-364TME can achieve conditional high-affinity activation at the tumor site. This enhances drug internalization and toxin release while significantly reducing off-target toxicity to normal tissues, providing a new path to solve the long-standing safety issues in the ADC field. Key preclinical research data show:
● Precise Targeting and Potent Killing: LM-364TME demonstrates an extremely wide binding window under simulated tumor microenvironment (high ANP) conditions, with efficient internalization, strong target-tumor cell cytotoxicity, and a significant bystander effect.
● Broad-Spectrum and Potent In Vivo Anti-tumor Activity: It has shown significant tumor inhibition in multiple PDX (patient-derived xenograft) models. Among them, the TGI (tumor growth inhibition) rate was 119.1% for triple-negative breast cancer, 107.46% for urothelial carcinoma, 168.79% for cervix carcinoma, and 86.73% for esophageal cancer.
● Safety: It was well-tolerated in 7-week repeat-dose toxicology studies in SD rats and rhesus monkeys. The HNSTD (Highest Non-Severely Toxic Dose) in cynomolgus monkeys reached 60 mg/kg.
Currently, LM-364TME has entered the US FDA IND submission stage and is expected to initiate its first-in-human clinical trial in 2026.
LM-338: An ADC Drug with Global First-in-Class (FIC) Potential

Sialyl-Thomsen-nouveau (STn) is a truncated O-glycan antigen that is virtually unexpressed in most normal tissues but highly expressed in various solid tumors (such as ovarian cancer, breast cancer, bladder cancer, cervix carcinoma, colorectal cancer, pancreatic carcinoma, and NSCLC), and is considered an ideal ADC target with great potential. LaNova Medicines's independently developed LM-338 is a potential global first-in-class ADC drug targeting STn, formed by precisely conjugating a humanized monoclonal antibody (LM-138) to a topoisomerase I inhibitor via a cleavable linker, with a drug-to-antibody ratio (DAR) of 4. Key preclinical research data show:
● High target specificity and internalization efficiency: Highly selective for the STn antigen, with extremely low cross-reactivity to the structurally similar sialic acid-T antigen, and possesses excellent internalization capability.
● Potent in vitro and in vivo anti-tumor activity: Demonstrated potent cytotoxicity and bystander effect in vitro; in multiple STn-positive cell line-derived xenograft (CDX) and PDX mouse models, including ovarian cancer, colorectal cancer, and NSCLC, LM-338 monotherapy induced significant tumor growth inhibition and even complete regression.
● Safety: Toxicology studies in rhesus monkeys showed an HNSTD of 60 mg/kg, with good overall tolerability.
These positive preclinical data have laid a solid foundation for the clinical development of LM-338 in STn-positive solid tumors.
From "conditional activation" to "highly selective targets", LaNova Medicines is systematically addressing the long-standing challenge of balancing safety and efficacy in the ADC field through two complementary pathways. As the two drug candidates, LM-364TME and LM-338, are about to enter the clinical development stage, LaNova Medicines is poised to bring a new generation of safer and more effective ADC treatment options to tumor patients worldwide.
Declaration:
1. This press release is intended to facilitate the communication and exchange of medical information and is for reference by healthcare professionals only. It is not for advertising purposes.
2. The company does not recommend any drugs and/or indications.
3. The information contained in this press release is for reference only and cannot replace professional medical guidance in any way, nor should it be considered as a diagnosis or treatment recommendation. If you wish to understand specific disease diagnosis and treatment information, please follow the advice or guidance of a physician or other healthcare professional.
Forward-Looking Statements:
This press release contains certain forward-looking statements, including statements regarding the clinical development plans, expected clinical benefits and advantages, commercialization prospects, potential for patient clinical benefit, and potential commercial opportunities for [LM-364TME, LM-338]. Words such as "expect", "believe", "continue", "may", "estimate", "hope", "intend", "plan", "potential", "predict", "project", "should", "will", "propose", and similar expressions are intended to identify forward-looking statements, but not all forward-looking statements contain these identifying words. These forward-looking statements are predictions or expectations made by the company based on currently available data and information, and actual results may differ materially from these forward-looking statements due to uncertainties or risks such as policy, R&D, market, and regulatory factors. Current or potential investors are advised to carefully consider the potential risks and should not place undue reliance on the forward-looking statements in this press release, which contain information only as of the date of this press release. Unless required by law, the company undertakes no obligation to update or revise any forward-looking statements in this press release as a result of new information, future events, or other circumstances.
- Previous:Major News! "Chinese Expert Consensus on the Diagnosis and Treatment of Chemotherapy-Induced Neutropenia (2026 Edition)" Released, Efbemalenograstim alfa Receives Authoritative Recommendation
- Next:AACR 2026 | LaNova Medicines Announces Preliminary Clinical Data for MK-2010/LM-299 for the First Time





